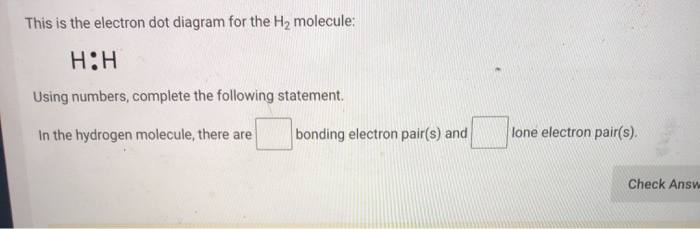
43 h2 electron dot diagram
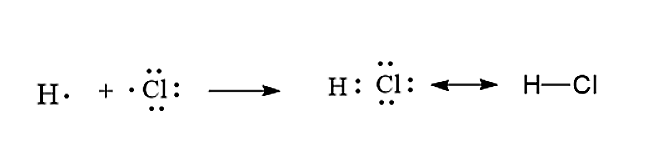
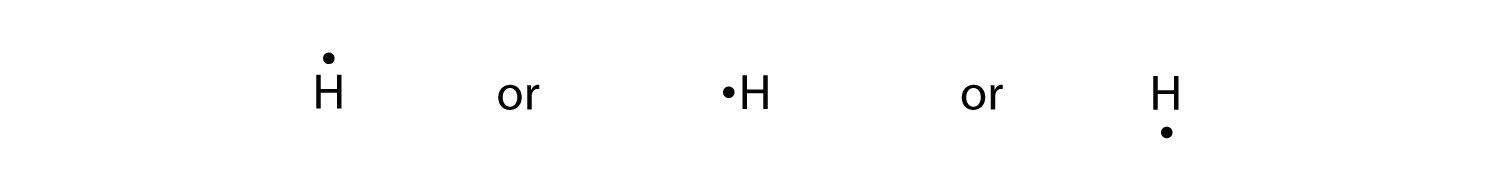
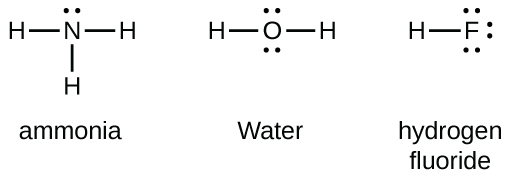
Consider the two electron arrangements for neutral atoms A and B. What is the difference between atom A and atom B? A - 1s2, 2s2, 2p6, 3s1 B - 1s2, 2s2, 2p6, 5s1 Atom B has lost some inner electrons. The outer electron of atom B has moved to a lower energy state. The outer electron of atom B has moved to a higher energy state. For example, a hydrogen molecule, H 2, forms when two hydrogen atoms each share their outer electron. A dot and cross diagram to show the bonding in hydrogen. An ammonia molecule, ...
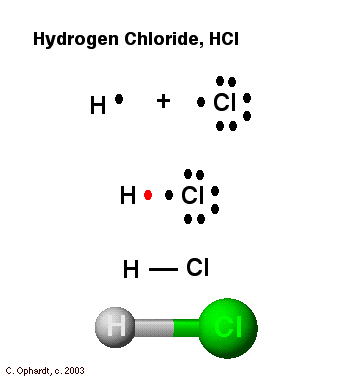
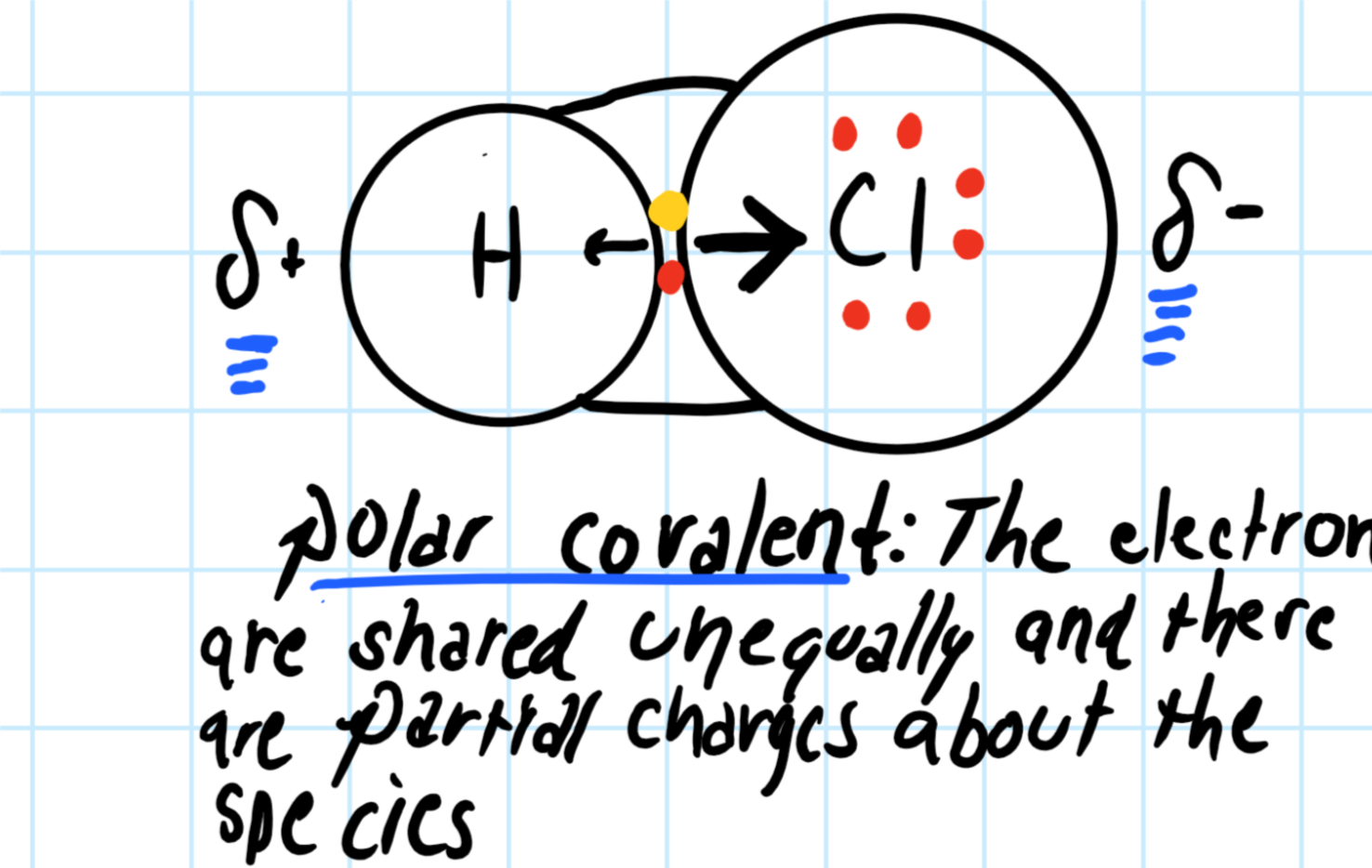
Each dot in the Electron dot structure will represent an electron. When electron dots are present in the pairs it will represent the chemical bond. - Hydrogen ...1 answer · Top answer: Hint: In order to draw an electron dot structure for hydrogen chloride molecule, we must first know what an electron dot structure is. Electron dot structure ...
/Lewis-dot-structure-58e5390f3df78c5162b4c3db.jpg)
H2 electron dot diagram
Enter the email address you signed up with and we'll email you a reset link. Jan 26, 2016 — H-H or H:H The hydrogen atom is happy when its valence shell has 2 electrons,So it shares 1 electron with the other hydrogen atom.1 answer · H−H or H:H Explanation: The hydrogen atom is happy when its valence shell has 2 electrons,So it shares 1 electron with the other hydrogen atom. (a) (i) Draw a dot and cross diagram of one molecule of hydrogen chloride. Show the outer electrons only. (ii) Explain why liquid hydrogen chloride has a low boiling point. (b) When dissolved in water, hydrogen chloride forms hydrogen ions (H+) and chloride ions (Cl–). (i) Draw a diagram of a chloride ion, showing the outer electrons only.
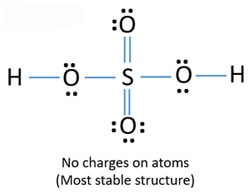
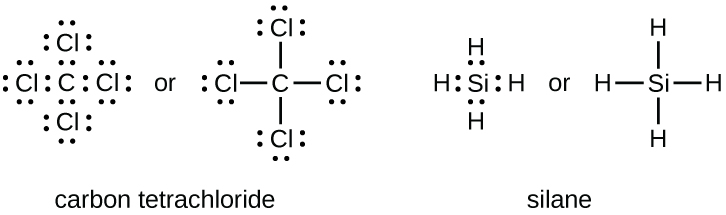
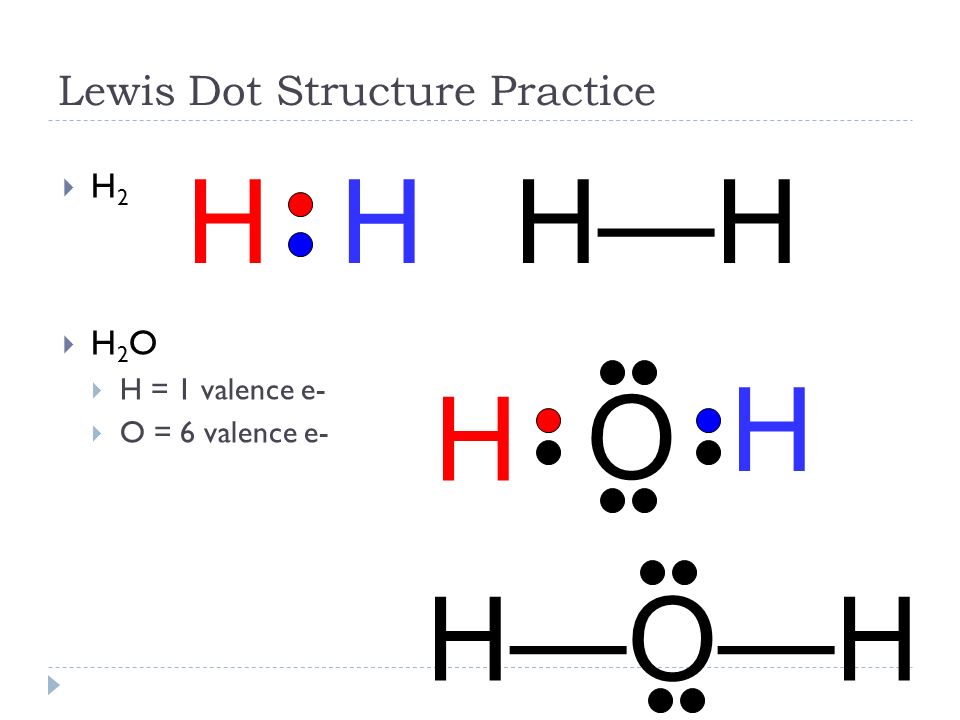
H2 electron dot diagram. May 27, 2020 — The electron dot structure or the Lewis Structure of H₂ shows how the two atoms of hydrogen share valence electrons and make a bond between them ...2 answers · 14 votes: Answer:Explanation:h2 has four electrons and the electron dot structure is here Image result for lewis dot of ccl4. Lewis strucuture of H2. Related image. Solve any question of Chemical Bonding and Molecular Structure with:- ...1 answer · Top answer: Lewis structure of H2O Lewis structure of CCl4 Lewis strucuture of H2 Based on the best Lewis electron-dot structure for SO42- and formal charge considerations, what is the predicted S-O bond order for each S-O bond? 1.5 Shown below is a model of PF5 having an orientation in which one or more atoms are hidden from view. Jan 30, 2022 · The Lewis Structure is therefore also known as electron dot structure and is one of the most predominant and simplest concepts to understand the chemical bonding of molecular compounds. Here, we will work with sulfate ion i.e SO42-. Step 1: Count the total number of valence electrons present in the molecule/ion
Dec 30, 2021 · The decay of the blue color of the electron in the TiO2 particle as well as the formation of the 484-nm absorption of [Co(C5H4COO)2]2- was used to monitor the course of the reaction. At pH 10, the electron spectrum has a max. at 780 nm and its extinction coeff. at this wavelength was detd. as 800/M-cm. (a) (i) Draw a dot and cross diagram of one molecule of hydrogen chloride. Show the outer electrons only. (ii) Explain why liquid hydrogen chloride has a low boiling point. (b) When dissolved in water, hydrogen chloride forms hydrogen ions (H+) and chloride ions (Cl–). (i) Draw a diagram of a chloride ion, showing the outer electrons only. Jan 26, 2016 — H-H or H:H The hydrogen atom is happy when its valence shell has 2 electrons,So it shares 1 electron with the other hydrogen atom.1 answer · H−H or H:H Explanation: The hydrogen atom is happy when its valence shell has 2 electrons,So it shares 1 electron with the other hydrogen atom. Enter the email address you signed up with and we'll email you a reset link.







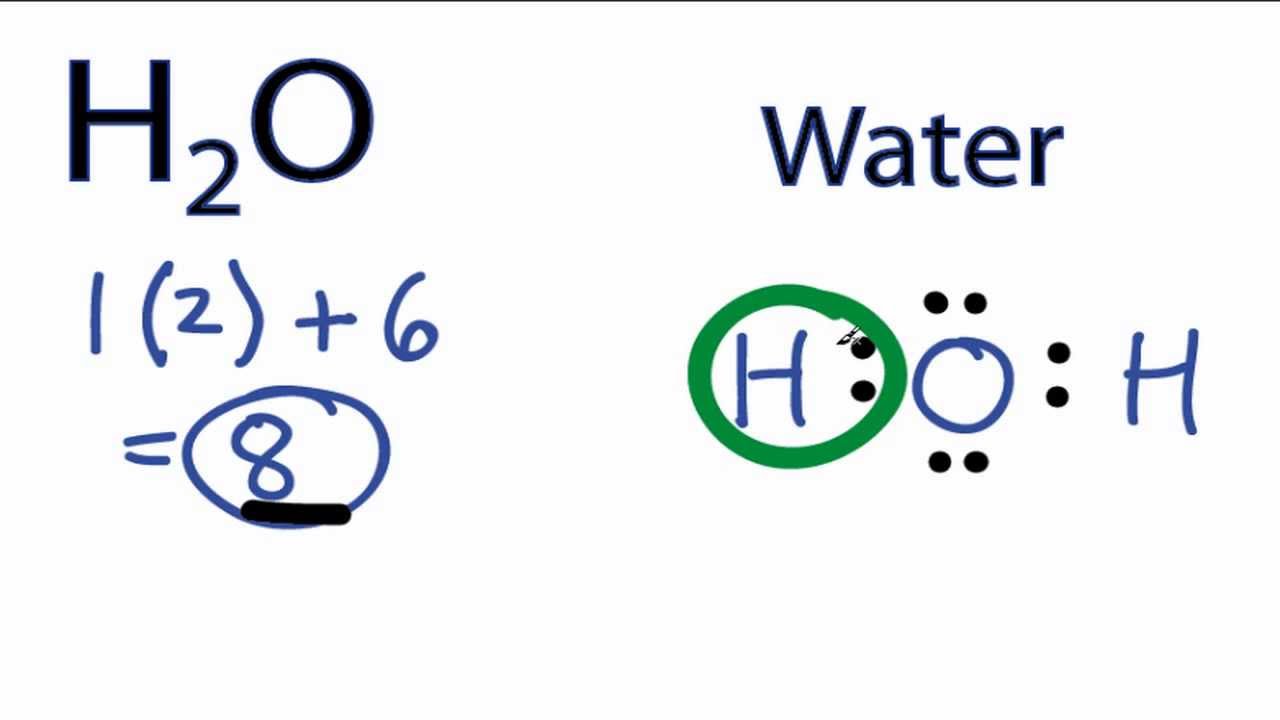
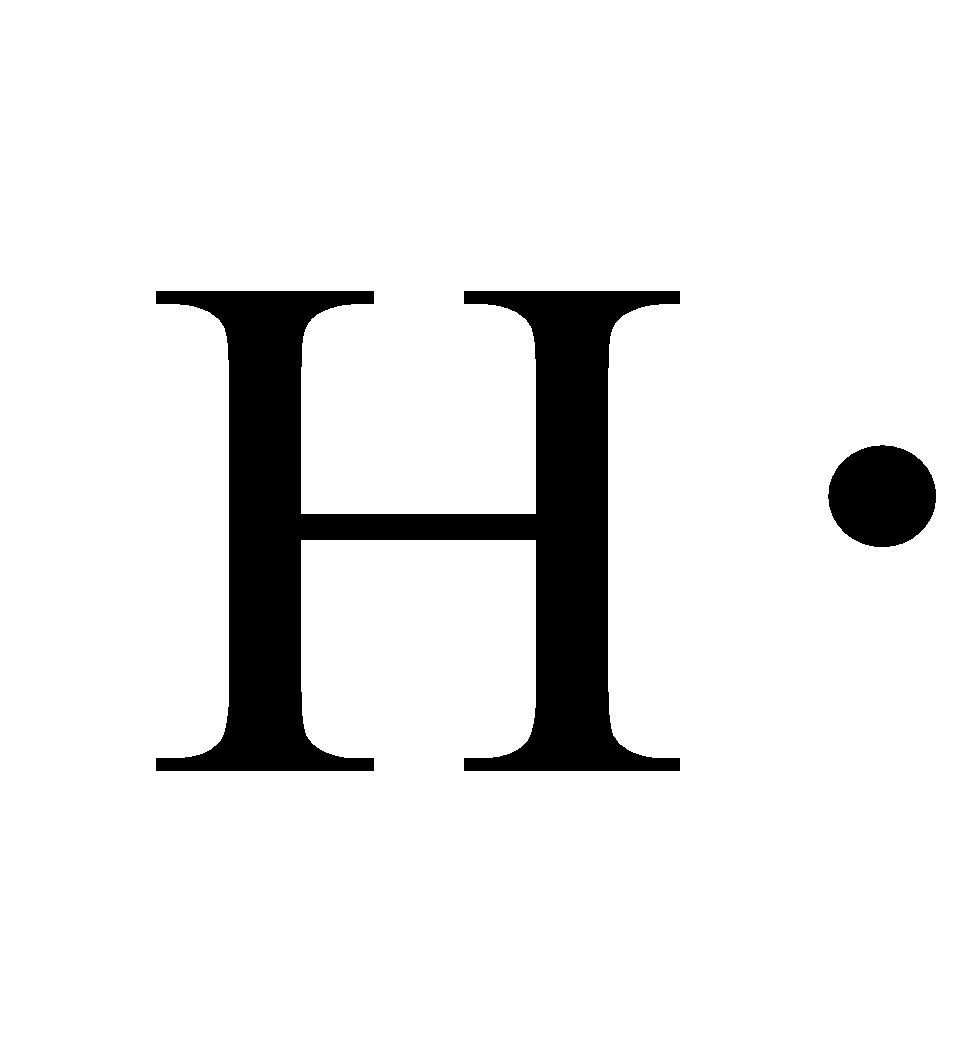



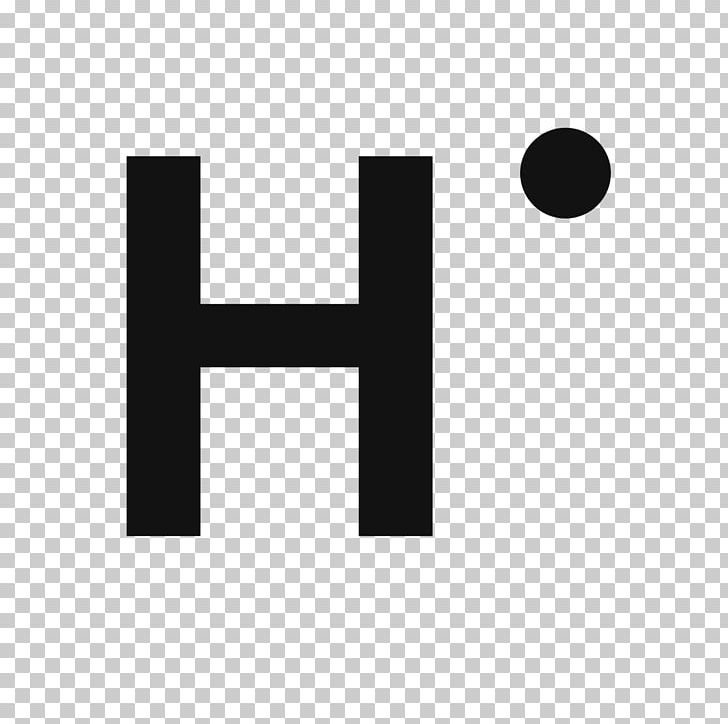



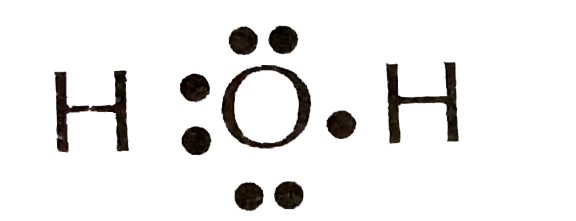










Comments
Post a Comment